Acute variceal hemorrhage is a serious cause of mortality in the emergency department and can be difficult to treat. In general, upper gastrointestinal (GI) hemorrhage accounts for 102 hospitalizations per 100,000 people every year, and esophageal varices represent approximately 14% of these cases.1,2 Furthermore, esophageal varices are the most common cause of persistent and severe upper GI hemorrhage, accounting for approximately 33% of these events.3 Among patients with cirrhosis, 70% of upper GI bleeding episodes are caused by esophageal varices.4 Gastroesophageal varices exist in nearly 50% of patients with cirrhosis at the time of initial diagnosis.5
The past 2 decades have seen significant advances in the treatment of variceal bleeding, but these events remain lethal in many cases. For initial bleeding episodes from esophageal varices, 6-week mortality stands at approximately 15%, down from 40% 25 years ago.6 This high mortality rate distinguishes variceal hemorrhage from other causes of upper GI bleeding, which often resolve spontaneously and have mortality rates below 3%.3 Given the lethality of this disease, it is more vital than ever for emergency physicians to understand current therapy for gastroesophageal varices and deliver life-saving treatment.
Pathophysiology
Although the basic pathophysiology of portal hypertension and variceal disease has been well established, research is ongoing to enable us to understand the mechanisms of this disorder and uncover important therapies. In the western world, portal hypertension is caused most often by cirrhosis and leads to portosystemic shunts and enlarged blood vessels at several anatomic locations, including the distal esophagus and proximal stomach.7 As portal pressure increases, these vessels become more likely to bleed. Worsening liver failure depletes coagulation factors, increases portal hypertension, and makes bleeding more likely.8 Importantly, bacterial infection also increases the risk of hemorrhage, particularly rebleeding.9
Two main prophylactic therapies are aimed at reducing these risk factors for hemorrhage. Beta-blocker therapy is recommended for use in cirrhotic patients with known esophageal varices, and acts to reduce portal hypertension, delay the advancement of varices, and prevent hemorrhage.5 In addition, the use of prophylactic antibiotics after an episode of variceal hemorrhage decreases the likelihood of rebleeding.10
Signs and Symptoms
Recognition of variceal hemorrhage in the emergency department is the first step toward proper management. Upper GI hemorrhage in patients with known liver disease should be treated as variceal bleeding until proven otherwise. The classic signs of upper GI hemorrhage are melena and hematemesis. Of patient presentations of melena or hematemesis, 5%-10% are caused by varices,11 but among patients with portal hypertension, ruptured varices account for 70% of all upper GI bleeding episodes.12,13
Several other signs and symptoms can be helpful in diagnosing variceal hemorrhage. Melena is typically associated with upper GI hemorrhage (proximal to the ligament of Treitz), but hematochezia may be a sign of brisk upper GI hemorrhage. Melena has been found to be present in 70%-80% of patients with upper GI hemorrhage, while hematochezia reportedly occurs in 15%-20% of these patients.3 Given these findings, all patients suspected of GI bleeding should have a rectal exam immediately. In addition, patients with liver disease and symptoms of anemia, including presyncope or syncope, fatigue, chest pain, or shortness of breath, should receive prompt evaluation for variceal hemorrhage.
Diagnostic Tests
Diagnosis of variceal hemorrhage often cannot definitively be made in the ED, but should be suspected on the basis of past medical history, history of present illness, and physical findings. GI hemorrhage in a cirrhotic patient should be assumed to be variceal until proven otherwise. In these patients, a number of diagnostic tests should be employed and may be useful in guiding treatment. Initial vital signs have been shown to have prognostic value. Specifically, shock (systolic blood pressure less than 100 mm Hg or heart rate greater than 100 bpm) confers a poorer prognosis for patients with upper GI bleeding, with mortality as high as 30% overall.3
When variceal bleeding is suspected, lab tests should include a complete blood count, comprehensive chemistry panel, coagulation studies, blood cultures, and troponin and lactate levels to assess the extent of the hemorrhage and its sequelae. A hemoglobin of less than 10 g/dL has been associated with a poor prognosis.3 The most important lab test, however, is a type and crossmatch for 2-6 units of blood, particularly in patients with large-volume hematemesis or other evidence of massive hemorrhage.3 An ECG to rule out related cardiac ischemia is worthwhile. One study found that nearly 50% of patients sent to the ICU for GI hemorrhage had laboratory evidence of cardiac damage.14
Gastric lavage via nasogastric tube traditionally has been used to assess if the bleeding is proximal to the duodenum and if the bleeding is active (as shown by clearing aspirate on repeated lavage). Although nasogastric aspiration has not been shown to improve outcomes, it has been linked to improved visual conditions for endoscopy.6,15,16 Endoscopic evaluation is the mainstay of diagnosis and treatment for evaluation of upper GI hemorrhage in general and variceal hemorrhage in particular. This should reinforce the importance of early consultation with GI specialists.
Treatment
As always, in critical conditions, the emergency physician should follow the airway, breathing, circulation sequence. Accordingly, the first priority in the management of variceal bleeding is to ensure a working airway. Early intubation should be considered for a number of reasons, including maintaining an airway with depressed mental status caused by hepatic encephalopathy, decreasing aspiration risk and hypoxia, and facilitating optimal endoscopy.12
Once the airway and breathing have been addressed, the circulation status will require a number of interventions. These patients should have intravenous access with two large-bore catheters or a central line to facilitate resuscitation. Volume resuscitation can begin with crystalloid solutions, but should include blood products in the case of active hemorrhage. Transfusion should be aimed at maintaining both hemoglobin at approximately 8 g/dL and hemodynamic stability.17
Although there is no recommended dosage of fresh frozen plasma or platelets, both should be considered in patients with marked coagulopathy or thrombocytopenia. Care should be taken while giving blood products, as overtransfusion can lead to worsening portal hypertension, and fresh frozen plasma and platelets have not been shown reliably to correct coagulopathy in these scenarios.7
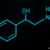
A number of pharmacologic interventions should be instituted for patients with variceal hemorrhage. Proton pump inhibitors (PPIs) have been prescribed for patients with suspected gastroduodenal ulcer disease, and should be considered in variceal hemorrhage, as well. Acid suppressive therapy is theorized to improve the stability of clot, and infusion of omeprazole has been shown to reduce risk of recurrent bleeding and need for emergent surgery in all cases of upper GI hemorrhage.11 Because it is difficult initially to differentiate esophageal variceal bleeding from ulcerative causes, all patients with upper GI hemorrhage should receive PPI infusion. Omeprazole and esomeprazole dosages are 80 mg IV bolus followed by 8 mg/hour IV infusion.11
Vasoactive drugs also have been shown to benefit these patients. Terlipressin, a vasopressin analog, has been shown to improve prognosis in variceal bleeding, preserving renal function and controlling hemorrhage. Unfortunately, it is not available in the United States. Octreotide has a demonstrated benefit for controlling hemorrhage, but it has not been shown to improve mortality on its own.12 It does, however, significantly decrease early rebleeding when used in combination with sclerotherapy, and it has found wide acceptance in the United States. Octreotide is given in a 50 mcg bolus followed by an infusion of 25-50 mcg/hour for 2-5 days.4
Antibiotic therapy decreases the release of endotoxins, which helps reverse systemic vasodilation, ameliorates worsening liver function and subsequent coagulopathy, and prevents early rebleeding.18 Most antibiotic regimens have been directed against gram-negative bacilli and have included drugs such as norfloxacin or ciprofloxacin. Ceftriaxone has been shown to be superior to quinolones and is recommended for 5-7 days of administration following a variceal hemorrhage.6
Another antibiotic, erythromycin, has been used primarily for its promotility effects, and has been shown to improve the quality of endoscopy, decrease time required to perform the endoscopy, and decrease the need for second-look procedures.19,20 Metoclopramide has also been used for this purpose, but erythromycin is likely more cost effective.21
Although these pharmacologic interventions are important, endoscopy remains the definitive treatment of choice for variceal hemorrhage. The details of endoscopic technique fall under the purview of gastroenterology, but it will be useful to examine general issues of timing here. Endoscopic band ligation (EBL) and sclerotherapy are the treatment modalities most frequently used, though EBL has been shown generally superior.22 For hemodynamically stable patients, early endoscopy has not been shown to improve outcomes over later endoscopic intervention.23 In stable patients, no significant differences have been shown between endoscopy within 6 hours of the bleeding event and within 24 hours.24 Early GI consultation remains beneficial for coordination of therapy, and potentially unstable patients should be seen urgently by GI specialists.
Some therapies that traditionally have been thought of as last-ditch rescue therapies, such as transjugular intrahepatic portosystemic shunt (TIPS) and portacaval shunt, are beginning to be used more widely. Recently, controlled trials have found that early TIPS or portacaval shunt significantly reduces treatment failure and mortality, particularly in patients at high risk for rebleeding.25 In one prospective trial, portacaval shunt procedures outperformed endoscopy in terms of prevention of rebleeding, prevention of encephalopathy, and long-term mortality.26 As evidence mounts of the safety and efficacy of TIPS in patients with less-severe liver failure, physicians treating variceal bleeding may begin considering it earlier in the course of treatment.
One treatment modality that likely will remain a salvage therapy is balloon tamponade. A rescue therapy for actively exsanguinating patients, the Sengstaken-Blakemore or Minnesota tube, when inflated within the esophagus, can control variceal bleeding in up to 90% of cases. The complication rate, however, approaches 30%, including aspiration pneumonia, airway obstruction, esophageal perforation, or mucosal injury. It is therefore recommended for use only by experienced clinicians in emergent situations, and after endotracheal intubation. Rebleeding following balloon deflation occurs in approximately 50% of cases.7
Recombinant factor VIIa has been studied as an adjunct therapy but so far has not shown sufficient utility to warrant routine use. Intended to improve coagulopathy during active bleeding, factor VIIa has not been shown to control bleeding or prevent rebleeding in randomized, controlled trials. There does appear to be an improvement in 6-week mortality. Given the cost involved and lack of firm evidence base, this drug should be used only as salvage therapy.6
Conclusion
Despite significant improvement in mortality in the last 25 years, variceal hemorrhage remains a deadly condition that can be difficult to manage in the emergency department setting. Keys to management include recognizing the poor prognosis and beginning treatment promptly, knowing the key pharmacologic interventions and coordinating care early with GI or surgical services as appropriate. One center found that prophylactic antibiotics were given to only 25% of variceal hemorrhage patients, while beta-blockers were given to only 24% of those with no contraindication, illustrating the need for improved communication and education.27 Informed emergency physicians are in an excellent position to make a difference in the care of these patients.
References
- Longstreth GF. Epidemiology of hospitalization for acute upper gastrointestinal hemorrhage. Am J Gastroenterol. 1995;20(2):206-10.
- Jutabha R and Jensen DM. Management of upper gastrointestinal bleeding in the patient with chronic liver disease. Med Clin North Am. 1996;80(5):P1035-67.
- Peter DJ and Dougherty JM. Evaluation of the patient with gastrointestinal bleeding. Emerg Med Clin North Am. 1999;17(1):239-61.
- Thabut D and Bernard-Chabert B. Management of acute bleeding from portal hypertension. Best Pract Res Clin Gastroenterol. 2007;21(1):19-29.
- Garcia-Tsao G and Bosch J. Management of varices and variceal hemorrhage in cirrhosis. N Engl J Med. 2010;362(9):823-32.
- Bendtsen F, et al. Treatment of acute variceal bleeding. Dig Liver Dis. 2008;40:328-36.
- Sass DA and Chopra KB. Portal hypertension and variceal hemorrhage. Med Clin North Am. 2009;93:837-53.
- Krige JEJ, et al. Predictive factors for rebleeding and death in alcoholic cirrhotic patients with acute variceal bleeding. World J Surg. 2009;33:2127-35.
- Goulis J, et al. Bacterial infection is independently associated with failure to control bleeding in cirrhotic patients with gastrointestinal hemorrhage. Hepatology. 1998;27:1207-12.
- Jun CH, et al. Antibiotic prophylaxis using third-generation cephalosporins can reduce the risk of early rebleeding in the first acute gastroesophageal variceal hemorrhage. J Korean Med Sci. 2006;21:883-90.
- Palmer K. Acute upper gastrointestinal hemorrhage. Br Med Bull. 2007;83:307-24.
- Abraldes JG and Bosch J. The treatment of acute variceal bleeding. J Clin Gastroenterol. 2007;41(3):S312-7.
- Bambha K, et al. Predictors of early re-bleeding and mortality after acute variceal hemorrhage in patients with cirrhosis. Gut. 2008;57:814-20.
- Bhatti N, et al. Myocardial infarction in critically ill patients presenting with gastrointestinal hemorrhage. Chest. 1998;114:1137-42.
- Witting MD, et al. Usefulness and validity of diagnostic nasogastric aspiration in patients without hematemesis. Ann Emerg Med. 2004;43(4):525-32.
- Lee SD and Kearney DJ. A randomized controlled trial of gastric lavage prior to endoscopy for acute upper gastrointestinal bleeding. J Clin Gastroenterol. 2004;38(10):861-5.
- Garcia-Tsao G, et al. Prevention and management of gastroesophageal varices and variceal hemorrhage in cirrhosis. Hepatology. 2007;46(3):922-38.
- Thalheimer U, et al. Infection, coagulation, and variceal bleeding in cirrhosis. Gut. 2005;54:556-63.
- Coffin B, et al.Erythromycin improves the quality of EGD in patients with acute upper GI bleeding. Gastrointest Endosc. 2002;56:174-9.
- Frossard JL, et al. Erythromycin intravenous bolus infusion in acute upper gastrointestinal bleeding. Gastroenterology. 2002;123:17-23.
- Winstead NS and Wilcox CM. Erythromycin prior to endoscopy for acute upper gastrointestinal haemorrhage. Aliment Pharmacol Ther. 2007;26:1371-7.
- Yan BM and Lee SS. Emergency management of bleeding esophageal varices: drugs, bands or sleep? Can J Gastroenterol. 2006;20(3):165-70.
- Cheung J, et al. Urgent vs. non-urgent endoscopy in stable acute variceal bleeding. Am J Gastroenterol. 2009;104:1125-9.
- Sarin N, et al. Time to endoscopy and outcomes in upper gastrointestinal bleeding. Can J Gastroenterol. 2009;23(7):489-93.
- Garcia-Pagan J, et al. Early use of TIPS in patients with cirrhosis and variceal bleeding. N Engl J Med. 2010;362(25):2370-9.
- Orloff MJ, et al. Randomized trial of emergency endoscopic sclerotherapy versus emergency portacaval shunt for acutely bleeding esophageal varices in cirrhosis. JACS. 2009;209:25-40.
- Singh H, et al. An assessment of endoscopic and concomitant management of acute variceal bleeding at a tertiary care centre. Can J Gastroenterol. 2007;21(2):85-90.




No Responses to “Variceal Hemorrhage”