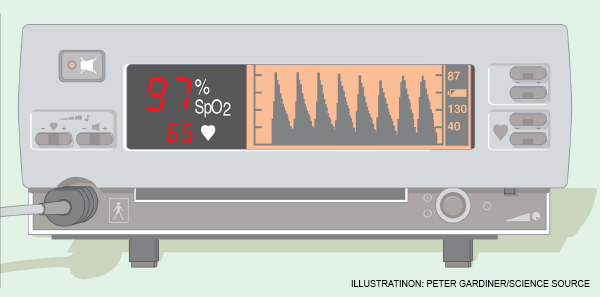
Procedural sedation and emergency airway management are recognized risks to patient safety. Sedation, induction agents, and muscle relaxants can quickly impact oxygen saturation, and desaturation is often precipitous.
Explore This Issue
ACEP Now: Vol 37 – No 04 – April 2018What contributes to the sorcery that seems to surround airway management and procedural sedation, and how can we avoid bad outcomes?
Gas Monitoring
First, recognize that with pulse oximetry, we use an imperfect monitor. It lags what’s happening in your patient by 30–90 seconds. Moreover, it does not give you an estimation of safe apnea time, even with high values (ie, 94–100 percent), because the asymptotic shape of the pulse oximeter curve displays saturation, not the amount of oxygen in the blood (ie, the partial pressure of O2 [PaO2] in a blood gas). A pulse oximeter reading of 95 percent may represent a PaO2 of 80, while a pulse oximeter reading of 100 percent can represent a PaO2 of anywhere from 95–600 (see Figure 1).
The argument for CO2 monitoring is that it monitors ventilation and therefore is immediate as well as forward looking. The problem: Most commonly used end-tidal CO2 detection methods (ie, small bore nasal cannulas) can’t provide higher flows (ie, >6 lpm) without popping off the oxygen source. Even if you rig a cannula or other mechanism to place the CO2 detector into or under a mask, high-flow oxygen given simultaneously affects CO2 detection.
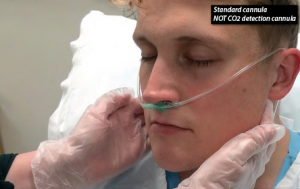
Figure 2: A standard nasal cannula, shown here, can be paired with a with a low-flow cannula with end-tidal CO2 detection to avoid affecting CO2 detection when higher flows are needed.
PHOTO: Richard Levitan
Perhaps the easiest way around this is to use two cannulas—that is, start with a low-flow cannula with end-tidal CO2 detection, and if you need higher flows, run oxygen through a standard nasal cannula previously placed on the patient (see Figure 2). Coming off the wall, a standard nasal cannula can deliver flows well above 50 lpm (even though the manometer only goes to 15 lpm) and as high as 70 lpm. “O’s up the nose” at these high flows can dramatically improve oxygenation, assuming the airway remains patent.
Divide the Airway
After more than twenty years of being airway obsessed, I recently began to gain a different perspective of the anatomy and clinical challenges of airway management. I believe it is useful to divide the airway into three sections to improve our anatomic understanding, and more importantly, to guide therapeutic intervention (see Figure 3):
- The upper airway includes the nasopharynx, mouth, and the hypopharynx down to the larynx. The upper airway is the most common site of airway obstruction due to the soft tissue structures of the palate, tongue, and epiglottis.
- The middle airway runs from the laryngeal cartilages (larynx) to the bronchi. It is normally patent, stented open by the rigidity of the thyroid and cricoid cartilage, and the tracheal rings.
- The lower airway includes the lungs and alveoli, where gas absorption occurs across the alveolar-capillary membrane.
To decipher the sorcery of the airway, we must appreciate how sedation, positioning, and our therapeutic interventions and techniques affect the airway at all three levels. Gravity is the enemy of both upper and lower airway patency when the patient is in a supine position. Supine positioning (coupled with poor muscular tone) causes the tongue to fall backwards against the soft palate and contact the posterior pharynx. Oral airways and/or nasopharyngeal airways are often needed to keep the soft palate and tongue from obstructing the airway. This is problematic because some patients may have respiratory depression or poor tone, but an oral airway may still trigger a gag response and vomiting, risking aspiration. Although mask ventilation techniques emphasize jaw thrust, struggling to maintain upper airway patency in a supine position is intrinsically self-defeating. It is also ergonomically difficult and frequently a multi-person task, especially in large patients.

Figure 3: Think of the airway in these three sections.
PHOTO: Richard Levitan
Supine positioning runs counter to lower airway (alveolar) patency. In the supine position, the lung’s upper areas compress the dependent alveoli, and abdominal contents push the diaphragm up, significantly reducing lung volume. Changing position from upright to supine reduces functional residual capacity (FRC) by as much as one liter. Because estimated FRC in adults measures approximately 2,400 ml, a reduction of 1,000 ml reduces FRC by 42 percent. Loss of tone (as occurs with anesthetic induction) or over-sedation further reduces FRC by 400 ml.
Collectively, then, FRC reduction from standing to supine plus loss of tone results in a reduction of roughly 52 percent! This reduction’s impact on alveolar gas absorption is dramatic, especially in patients who already have compromised lung function.
Sit Them Up
The first and most important technique for boosting oxygen absorption is to apply high oxygen concentration with high flow as a means of augmenting the patient’s negative inspiratory efforts. But beyond the application of 100 percent oxygen, and shy of extracorporeal membrane oxygenation or using hyperbaric oxygenation, clinicians have only two methods of further improving oxygenation across the alveoli: 1) positioning, and 2), positive end-expiratory pressure (PEEP).
The immediate and easiest positioning fix to open the lower airway? Sit the patient up. This causes the abdominal contents to move caudad, the diaphragm to drop, and lung volume to rapidly expand. The area for gas absorption across the alveioli dramatically increases with proper positioning.
Middle airway obstruction is an infrequent clinical problem because the three-dimensional shape of laryngeal cartilages and the cartilaginous rings of the trachea maintain patency regardless of positioning or muscular tone. An orally placed tracheal tube traverses the upper airway and reaches the mid-trachea level. When the middle airway obstructs due to intrinsic laryngeal-tracheal pathology (eg, tumors, angioedema about the larynx, blood clots, etc.) or trauma (eg, laryngeal fracture, direct tracheal injury, etc.), patients can die precipitously.
With an awareness of gravity and positioning, and an understanding of the three airway components, engineering airway interventions that augment patency and gas absorption makes sense. Here’s my stepwise approach to hypoxia with procedural sedation or in an initial emergency department presentation:
- Sit the patient up, allowing the diaphragm to drop and the alveoli volume to expand, and pull on the mandible. This opens the upper airway.
- Send O’s up the nose—blast open the soft palate and shoot oxygen into the trachea up to and beyond 15 lpm.
- Add PEEP with a bag valve mask (BVM) and a PEEP valve, or a continuous positive airway pressure system. PEEP is absolutely necessary when high-flow oxygen (with cannula plus mask) does not achieve high oxygen saturations (ie, >98 percent).
To desaturate after the tracheal tube has been placed, first examine the system to ensure the tube is at right depth, the oxygen is connected, and the cuff is up. Suction through the tube to remove mucous plugs, clots, etc. If high-pressure alarms go off or bagging difficulty persists (that is not consistent with the patient’s pathology, ie, COPD, asthma, etc.), pull the tracheal tube. It is quite common to experience ball-valve obstructions from mucous plugs or clots that suctioning alone doesn’t resolve.
Plan to maximize oxygenation in every instance of procedural sedation. Use gravity to reduce the risk of aspiration, and always ask, “Do you need the patient flat?” We rarely do, and head elevation adds tremendously to patient safety.
The highest-risk patients may be those sedated for hip reduction because they need to be lying flat. Even a slight amount of head elevation (ie, 10 degrees) tilts the pannus down and improves lung function tremendously. Once the hip reduction is complete, bring the head higher and pull on the mandible with the nasal cannula on. Often the most dangerous part of sedation occurs after the reduction because pain input drops but the drugs haven’t worn off.
If positioning, pulling on the mandible, and nasal oxygen at high flow does not resolve hypoxia, add PEEP by using a BVM with a PEEP valve. Install PEEP valves on every BVM in your department so when PEEP is needed, you don’t need to locate one.
Be mindful of patients at high risk of desaturation due to alveolar disease (ie, heart failure, acute respiratory distress syndrome, multi-lobar pneumonia, aspiration, etc.). The sickest of the sick may require combining both upright positioning and PEEP for pre-oxygenation, and also during the onset phase of muscle relaxation when performing rapid sequence intubation. Even after successful intubation, these patients can prove difficult to oxygenate. Increase the fraction of inspired O2, increase PEEP, lower tidal volumes, and increase the ventilator rate in these challenging cases.
Finally, consider prone positioning if upright positioning doesn’t allow for sufficient oxygenation.
Pages: 1 2 3 | Multi-Page






No Responses to “Tips to Improve Airway Management”