The Case
A 61-year-old healthy female presents with two hours of right-side weakness as well as slurred speech. She has a National Institutes of Health Stroke Score/Scale (NIHSS) of 10, and you are concerned about a large vessel occlusion (LVO) based on the high NIHSS as well as the presence of both an upper extremity drift and the speech abnormality. A noncontrast computed tomography (CT) scan of her head shows no evidence of a bleed. A CT angiogram plus CT perfusion demonstrate a clot in the left proximal middle cerebral artery (MCA), with a small infarcted area and a large penumbra. She is a candidate to receive endovascular therapy (EVT) based on your institution’s current guidelines, but she is also within the current window for the administration of tissue plasminogen activator (tPA).
Clinical Question
Should you give tPA while waiting for your neurointerventional team to arrive, or is EVT alone noninferior to EVT with tPA?
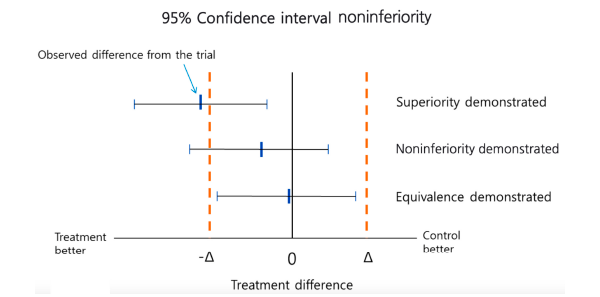
Figure 1: Testing Superiority and Equivalence/Noninferiority
Korean J Pediatr. 2012;55(11):403-407. CC BY-NC
Background: Thrombolytics as a possible treatment for acute ischemic stroke (AIS) has been discussed for decades. The famous NINDS trial led to tPA being approved for AIS within three hours of onset in those patients who met strict criteria.1 ECASS III was published in 2008 and expanded the window to 4.5 hours.2 ACEP has published guidelines on the safety and efficacy of tPA for patients with AIS. The ACEP guidelines give level B and level C recommendations.3
The controversy over the use of tPA for AIS does not seem to be going away. A recent reanalysis of ECASS III failed to show the commonly assumed benefit but confirmed the potential increase in harm of tPA. 4,5
The treatment of AIS did substantially change with the publication of the MR CLEAN trial.6 This was the first study demonstrating a benefit to endovascular treatment of a specific subset of ischemic stroke patients—specifically, those with large vessel occlusions presenting within six hours of symptom onset. MR CLEAN was followed by a number of other publications seeking to replicate and refine treatment as well as expand the window for treatment.
One major question regarding EVT treatment was whether the use of systemic thrombolytics like tPA results in better outcomes or if it simply exposes the patient to increased risks at a higher cost.
Reference: Yang P, Zhang Y, Zhang L, et al. Endovascular thrombectomy with or without intravenous alteplase in acute stroke. N Engl J Med. 2020;382(21):1981-1993.
- Population: Patients 18 years of age or older presenting within 4.5 hours of AIS symptoms with a cerebral vascular occlusion on CT angiography of the intracranial internal carotid artery or middle cerebral artery (first and/or second segments) and an NIHSS >1, and if endovascular thrombectomy was intended to be performed.
- Exclusions: Disability from a previous stroke or contraindication to IV alteplase, plus any contraindication for thrombolysis according to American Heart Association guidelines.
- Intervention: Endovascular thrombectomy.
- Comparison: Endovascular thrombectomy plus systemic tPA (0.9 mg/kg).
- Outcomes:
- Primary Outcome: Modified Rankin Scale (mRS) score assessed at 90 days.
- Secondary Outcomes: Death from any cause at 90 days, successful reperfusion before thrombectomy, recanalization at 24–72 hours, NIHSS score at 24 hours and five-to-seven days, and final lesion volume on CT and mRS comparisons.
- Safety Outcomes: All hemorrhages and symptomatic intracranial hemorrhages, occurrence of pseudoaneurysm and groin hematoma at the site of arterial puncture used for thrombectomy, cerebral infarction in a new vascular territory at five to seven days, and mortality within 90 days.
Authors’ Conclusions
“In Chinese patients with acute ischemic stroke from large-vessel occlusion, endovascular thrombectomy alone was noninferior with regard to functional outcome, within a 20 percent margin of confidence, to endovascular thrombectomy preceded by intravenous alteplase administered within 4.5 hours after symptom onset.”
Table 1: Secondary Outcomes
| Thrombectomy Alone | Combination Therapy | Relative Risk (95% Confidence Interval) | |
|---|---|---|---|
| Mortality at 90 days | 0.177 | 0.188 | 0.94 (0.68–1.30) |
| Symptomatic intracranial hemorrhage | 0.043 | 0.061 | 0.70 (0.36–1.37) |
| Successful reperfusion prior to endovascular therapy | 2.4% | 0.07 | 0.33 (0.14–0.74) |
| Overall successful reperfusion | 0.794 | 0.845 | 0.70 (0.47–1.06) |
| Recanalization at 24–72 hours | 0.851 | 0.891 | 0.71 (0.42 to 1.20) |
Key Results
The study included 656 patients with a median age of 69 years, and slightly more were male. The median NIHSS score was 17.
- Primary Outcome: Adjusted odds ratio (aOR) for the mRS.
- aOR = 1.07 (95% CI, 0.81–1.40), demonstrating noninferiority because the lower limit of noninferiority was set at 0.80.
- Secondary Outcomes: see Table 1.
Evidence-Based Commentary
1) Consecutive Patients: The publication did not explicitly say patients were recruited consecutively. Without this information, it is hard to comment on whether there was selection bias.
2) Blinding: There was a lack of blinding of the treating physicians and study participants. This could have biased the study toward the EVT alone if that hypothesis was known to these two groups.
3) Intention-to-Treat (ITT): Using an ITT analysis is a quality indicator for superiority designs. However, for noninferiority trials, a per-protocol analysis is the more conservative approach to minimize bias (see Figure 1). Using an ITT can bias the results toward the null hypothesis (ie, finding noninferiority). The authors did perform a per-protocol analysis, but this could only be found in the supplemental material.
4) Patient Outcomes: These data were obtained by interviews either performed in person or by telephone. The authors did not provide information on how many assessments were performed using each method. Telephone interviews are suboptimal for assessing functionality. It would have been helpful in interpreting the data to see if there were any differences that could be attributed to the way outcomes were assessed. Wider confidence intervals around the point estimate of efficacy could have been introduced into the data set by telephone interviews and biased the results toward finding noninferiority.
5) External Validity: The standard AIS care in China may contain differences compared to the United States, limiting the external validity of these results.
Bottom Line
If EVT is readily available and the patient is a candidate for this treatment modality, there does not appear to be a role for systemic thrombolysis in patients with acute ischemic stroke.
Case Resolution
You engage in shared decision making with the patient and her spouse. The decision is to hold off on administration of systemic thrombolytics and await the neurointerventional team. They arrive and take the patient for EVT, and she has a successful outcome.
Thank you to Dr. Anand Swaminathan, an assistant professor of emergency medicine at St. Joseph’s Regional Medical Center in Paterson, New Jersey, for his help with this review.
Remember to be skeptical of anything you learn, even if you heard it on the Skeptics’ Guide to Emergency Medicine.
References
- National Institute of Neurological Disorders and Stroke rt-PA Stroke Study Group. Tissue plasminogen activator for acute ischemic stroke. N Engl J Med. 1995;333(24):1581-1587.
- Hacke W, Kaste M, Bluhmki E, et al. Thrombolysis with alteplase 3 to 4.5 hours after acute ischemic stroke. N Engl J Med. 2008;359(13):1317-1329.
- American College of Emergency Physicians Clinical Policies Subcommittee (Writing Committee) on Use of Intravenous tPA for Ischemic Stroke: Brown MD, Burton JH, Nazarian DJ, et al. Clinical policy: use of intravenous tissue plasminogen activator for the management of acute ischemic stroke in the emergency department. Ann Emerg Med. 2015;66(3):322-333.e31.
- Milne WK, Lang E, Ting DK, et al. CJEM debate series: #TPA should be the initial treatment in eligible patients presenting with an acute ischemic stroke. CJEM. 2020;22(2):142-148.
- Alper BS, Foster G, Thabane L, et al. Thrombolysis with alteplase 3-4.5 hours after acute ischaemic stroke: trial reanalysis adjusted for baseline imbalances. BMJ Evid Based Med. 2020;bmjebm-2020-111386.
- Berkhemer OA, Fransen PSS, Beumer D, et al. A randomized trial of intraarterial treatment for acute ischemic stroke. N Engl J Med. 2015;372(1):11-20.
Pages: 1 2 3 | Multi-Page





No Responses to “Endovascular Therapy With or Without tPA—What Do the Studies Say?”