End-tidal capnography has gained momentum over the years as a standard for monitoring patients undergoing procedural sedation in the emergency department, with a level B recommendation coming out of ACEP’s clinical policy regarding procedural sedation in 2014.1 It can identify hypoventilation earlier than other monitoring tools we have at our disposal in the emergency department, but its utility doesn’t end there. It can quickly and efficiently answer clinical questions beyond that of sufficient ventilation. Are the chest compressions being performed on your cardiac arrest inadequate? Should you stop resuscitation efforts? Is your hyperglycemic diabetic in diabetic ketoacidosis (DKA)? Is that nasogastric tube in the stomach? End-tidal capnography can lend insight to these questions that emergency physicians encounter on a daily basis. End-tidal carbon dioxide (EtCO2) sensibly correlates with the pathophysiology of those and many other disease processes and can help guide decision making on your next shift.
Explore This Issue
ACEP Now: Vol 35 – No 11 – November 2016Capnography offers an indirect method to detect metabolic acidosis. EtCO2 measurements have been shown to closely estimate arterial partial pressure of carbon dioxide (pCO2) in healthy patients and also in the presence of metabolic derangements such as acidosis.
Understanding the Capnogram
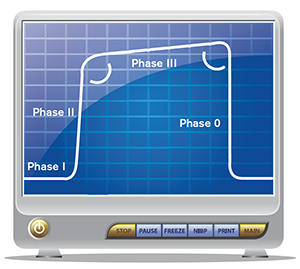
Figure 1. Normal, healthy capnogram.
The end-tidal capnogram is separated into four separate phases (see Figure 1). Phase 0 begins during the inhalation phase of the respiratory cycle and the capnogram drops precipitously from its peak level at the end of expiration. Once the patient begins to exhale (phase I), the initial expired air is predominantly dead space with little expired carbon dioxide (CO2), but as the more densely concentrated CO2 is expired, there is a sharp increase in the end-tidal waveform that represents phase II. The waveform then plateaus during phase III, with slight increases in the CO2 concentration from alveolar air. The discrete end-tidal number we refer to is the value at the end of phase III, the very end of expiration prior to inhaling the next breath. Depending on the situation, the actual waveform of the capnogram and/or the end-tidal value at the end of exhalation may help us with more than just determining hypoventilation in patients undergoing procedural sedation in the emergency department. (See our September 2015 article, “How to Use End-tidal Capnography to Monitor Asthmatic Patients,” at www.acepnow.com/article/how-to-use-end-tidal-capnography-to-monitor-asthmatic-patients for more information on using capnography and for a detailed review on using capnography to evaluate asthmatic patients.)
A New Breathalyzer for Detecting DKA?
It’s nearing the end of your shift when you sign up for your last patient of the day. This fast-breathing diabetic had been sitting in Fast Track for a leg infection, and when the glucometer flashed “HIGH,” he was moved to your higher-acuity area for more workup. You cringe as you picture the needle puncturing his wrist for an arterial blood gas (ABG) test and wonder whether you’ll be breaking out that insulin drip. DKA is an endocrine emergency hallmarked by hyperglycemia, ketonemia, and metabolic acidosis. Ma et al demonstrated that knowledge of the arterial pH in suspected DKA added little to clinical gestalt and altered the emergency physician’s management in only 2.5 percent of cases.2 Do we even need to subject our patients to this test? Although the pH of the often less painful venous blood gas has been shown to be comparable to that of an arterial blood gas, you wish you had an even quicker noninvasive screening tool.3
Capnography offers an indirect method to detect metabolic acidosis. EtCO2 measurements have been shown to closely estimate arterial partial pressure of carbon dioxide (pCO2) in healthy patients and also in the presence of metabolic derangements such as acidosis. Bou Chebl et al illustrated that lower pCO2 values correspond to lower pH and bicarbonate values in hyperglycemic diabetic patients, which is in line with the known pathophysiology of acid-base disturbances.4 In more severe cases of underlying metabolic acidosis, one would expect an increase in compensatory tachypnea and, therefore, lower EtCO2 readings. But how low buys you an insulin drip and a costly ICU bed? According to recent data (see Table 1) among patients with screening Accu-Cheks greater than 550 mg/dL, an EtCO2 of 35 or greater virtually guarantees that the patient is not in DKA with a sensitivity of 100 percent.4 On the other hand, EtCO2s of ≤21 and ≤26 are 100 percent and 96 percent specific for DKA, respectively.4 Among patients with a blood glucose (BG) >250 mg/dL, an EtCO2 greater or less than 24.5 is both 90 percent sensitive and 90 percent specific for DKA.5 In a pediatric population with hyperglycemia, similar cutoff points were delineated: an EtCO2 of >29 and ≥36 were 83 percent and 100 percent sensitive in ruling out DKA, respectively, while an EtCO2 of 6.
There is no current consensus regarding which EtCO2 levels can be used consistently in practice to rule in or rule out metabolic acidosis, and therefore DKA, in the right clinical setting; however, utilizing end-tidal capnography in the evaluation of diabetic patients with suspected DKA is a quick and noninvasive method both to approximate the presence and severity of metabolic acidosis and to guide your initial treatment and the patient’s ultimate disposition. While no formal consensus exists, a screening EtCO2 of ≥36 in your next hyperglycemic diabetic patient can rule out ketoacidosis and spare your patient an invasive ABG test.
End-Tidal to Monitor COPD Exacerbations
People across the world continue to smoke despite the best efforts of the Truth campaign. COPD exacerbations seem as prominent as ever among ED visits and thus lead to difficult clinical decisions regarding treatment plans, intubation, and disposition. Many emergency physicians rely on ABG analysis in conjunction with their physical assessments to make the appropriate disposition for patients with COPD exacerbation. Could EtCO2 also be the answer for COPD or just another far-fetched fairy tale?
Just like in most fairy tales, some things are just too good to be true. In a 2012 study, Soleimanpour et al were able to show a strong correlation between EtCO2 and pCO2 in normal healthy adults, with only a small discrepancy of 2 to 5 mmHg less in EtCO2 as compared to pCO2 on ABG.8 In another study by Yosefy et al, EtCO2 successfully predicted pCO2 in emergency department patients in respiratory distress; however, these studies were conducted and proved a strong correlation between EtCO2and pCO2 in “normal, healthy” patients.9 In additional studies of capnography use in respiratory patients, researchers have found the correlation between EtCO2 and pCO2 is not nearly as strong when the patient has shunting or mismatch perfusion as a result of underlying pulmonary disease. It is theorized that the increased dead-space ventilation creates an increasing gradient among EtCO2 and pCO2, therefore changing the correlation between one another to a mere moderate level. In a 2011 study by Kartal et al, the agreement between EtCO2 and pCO2 in COPD patients was a discrepancy of 8.4 mmHg less in EtCO2 as compared to pCO2.10 A similar study in COPD patients in 2015 was conducted by Taghizadieh et al and showed an even greater discrepancy among EtCO2 and pCO2, as much as 23 mmHg less in EtCO2 compared to arterial pCO2.11 Based on these small clinical trials looking specifically at COPD patients, the EtCO2 value is only minimally to moderately useful as a screening tool for hypercapneic respiratory failure. However, EtCO2 seems to remain consistently below pCO2 and may provide a starting point to monitor for improvement or worsening CO2 retention. Unlike asthmatics, the numerical end value of the EtCO2 capnogram can be used in your next COPD exacerbation patient to trend improvement or worsening respiratory failure, keeping in mind the correlation between EtCO2 and pCO2 is far from perfect in COPD patients.
In the next “Tricks of the Trade” column, we’ll review how to use end-tidal capnography to check orogastric/nasogastric tube placement and guide cardiopulmonary resuscitation.
 Dr. D’Amore is an emergency medicine resident at St. Joseph’s Regional Medical Center in Paterson, New Jersey.
Dr. D’Amore is an emergency medicine resident at St. Joseph’s Regional Medical Center in Paterson, New Jersey.
References
- Godwin SA, Burton JH, Gerardo CJ, et al. Clinical policy: procedural sedation and analgesia in the emergency department. Ann Emerg Med. 2014;63(2):247-258.
- Ma OJ, Rush MD, Godfrey MM, et al. Arterial blood gas results rarely influence emergency physician management of patients with suspected diabetic ketoacidosis. Acad Emerg Med. 2003;10(8):836-841.
- Byrne AL, Bennett M, Chatterji R, et al. Peripheral venous and arterial blood gas analysis in adults: are they comparable? A systematic review and meta-analysis. Respirology. 2014;19(2):168-175.
- Bou Chebl R, Madden B, Belsky J, et al. Diagnostic value of end tidal capnography in patients with hyperglycemia in the emergency department. BMC Emerg Med. 2016;16:7.
- Soleimanpour H, Taghizadieh A, Niafar M, et al. Predictive value of capnopgraphy for suspected diabetic ketoacidosis in the emergency department. West J Emerg Med. 2013;14(6):590-594.
- Fearon DM, Steele DW. End-tidal carbon dioxide predicts the presence and severity of acidosis in children with diabetes. Acad Emerg Med. 2002;9(12):1373-1378.
- Gilhotra Y, Porter P. Predicting diabetic ketoacidosis in children by measuring end-tidal CO2 via non-invasive nasal capnography. J Pediatr Child Health. 2007;43(10):677-680.
- Soleimanpour H, Gholipouri C, Golzari SEJ, et al. Capnography in the emergency department. Emerg Med. 2012; 2(9):e123.
- Yosefy C, Hay E, Nasri Y, et al. End tidal carbon dioxide as a predictor of the arterial PCO2 in the emergency department setting. Emerg Med J. 2004; 21(5):557-559.
- Kartal M, Goksu E, Eray O, et al. The value of ETCO2 measurement for COPD patients in the emergency department. Eur J Emerg Med. 2011;18(1):9-12.
- Taghizadieh A, Rahmani F, Soleimanpour H, et al. Comparison of end tidal carbon dioxide and arterial blood bicarbonate levels in patients with exacerbation chronic obstructive pulmonary disease. Thrita. 2015;4(2):e26169.
Pages: 1 2 3 4 | Multi-Page




No Responses to “End-Tidal Capnography Can be Useful for Detecting Diabetic Ketoacidosis, Monitoring COPD”