More than 6 million emergency department visits a year in the United States are for chest pain.1 While there are approximately 1.1 million hospitalizations a year for acute coronary syndrome (ACS) in the United States, only approximately 30 percent of cases are ST-elevation myocardial infarctions.2 The remaining 70 percent are diagnosed with unstable angina (UA) or non–ST-elevation myocardial infarction (NSTEMI). Risk stratification of chest pain patients with concern for ACS is of high concern in the emergency department. The American Heart Association (AHA) current guidelines recommend prompt management and possibly invasive strategies for patients with UA and NSTEMI who present with high-risk features.3 Often in the ED setting, chest pain patients have a nondiagnostic electrocardiogram (ECG), and initial cardiac biomarkers can be negative even with significant coronary artery occlusion.
Explore This Issue
ACEP Now: Vol 34 – No 02 – February 2015Novice sonographers may have difficulty obtaining clear views and should not base decisions on suboptimal imaging. Even for the experienced ED sonographer, differentiating between new and old RMWAs can be extremely challenging.
Investigation of regional wall motion abnormalities (RWMAs) is a Class I recommendation by the AHA in the hands of trained echocardiogram technicians.4 The goal is to identify patients with RWMAs likely representing a significant occult coronary artery thrombosis not evident by symptoms, ECG, or initial cardiac biomarkers that could then benefit from an invasive intervention. Previous studies suggest that if a RWMA is present, a large area of myocardium is at risk for death.5–7 Initial studies have shown good sensitivity and specificity of the identification of RWMAs for coronary ischemia in the ED setting.8,9 Most of these studies were performed by trained ECG technicians or cardiologists. However, several articles recently described emergency physicians of various levels of training being capable of identifying RWMAs.10,11 A case series described three cases in which emergency physicians identified RWMAs in patients with equivocal ECGs; all the cases went to cardiac catheterization partly based on the point-of-care echocardiography (POC echo) detecting RWMA and were found to have significant single-vessel coronary disease requiring intervention.12
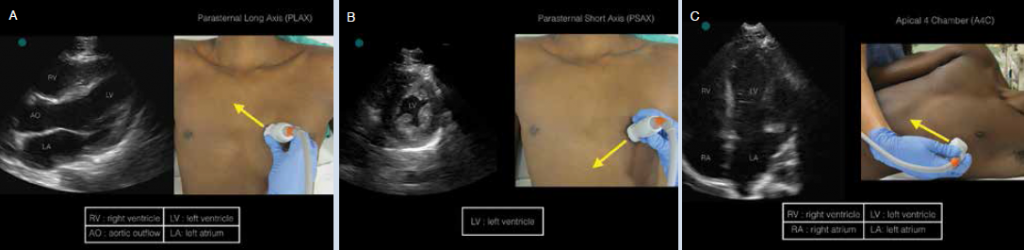
(click for larger image) Figure 1: Appropriate probe positioning and corresponding ultrasound image of A) PLAX, B) PSAX, and C) A4C.
Clinical Indications for Performing POC Echo for RWMAs
Evaluation for RWMAs should promptly occur when the emergency physician has a high concern for UA or NSTEMI by history and physical examination with an equivocal ECG for cardiac ischemia.
Anatomy
For simplicity, the traditional 17-wall motion segment identification on ECG has been

(click for larger image) Figure 2: Anatomical description of coronary perfusion in the A) PLAX, B) PSAX, and C) A4C.
condensed into a modified three-area evaluation that corresponds roughly to the major coronary artery perfusion territories of the left ventricle. This is based on American Society of Echocardiography guidelines and cardiac magnetic resonance imaging data of patients with acute coronary ischemia and has been successfully utilized in prior ED studies.12–14 Visualization of the left ventricle in PLAX, PSAX, and A4C can roughly display RWMAs of the three major coronary arteries: left anterior descending (LAD), circumflex (Cx), and right coronary artery (RCA) (see Figure 2). Thus an anterior RWMA corresponds with an occlusion of the LAD, a lateral RWMA corresponds with an occlusion of the Cx artery, and an inferior RWMA corresponds with an occlusion of the RCA.
Findings
Left ventricular RWMA is described as a hypokinesis, dyskinesis, or akinesis of a segment when compared to the other contracting segments of the chamber. This can be visualized sonographically as a blunting of the typical symmetric myocardial thickening during contraction as compared to other cardiac wall segments. This abnormal movement of the wall segment is highly suspicious for an associated coronary thrombus.

(click for larger image) Figure 3: Echocardiogram images with outlined regional wall motion abnormality territories of A) PSLAX B) PSAX, and C) A4C.
The anterior wall is supplied by the LAD, whereas the lateral wall is supplied by the Cx, and the inferior wall is supplied by the RCA (PLAX view, see Figure 3a). In our opinion, the PSAX at the level of the papillary muscles is often the best ultrasound view to appreciate RWMAs (see Figure 3b). In the A4C view, the proximal intraventricular septum is supplied by the RCA, while the rest of the visualized myocardium is supplied by the LAD (see Figure 3c). All three major vessels are roughly equally distributed over the left ventricle. However, some variation is to be expected.
ECG Correlation
Correlation of the ECG and POC echo images is helpful when attempting to determine the presence of RWMAs (see Figure 4). An anterior RWMA (LAD distribution) should correlate with ECG changes in V1 and V2. A lateral RWMA (Cx distribution) should correlate with ECG changes in V5, V6, I, and aVL. An inferior RWMA (RCA distribution) should correlate with ECG changes in II, III, and aVF.
Next Steps in Management
An identified RWMA can often suggest a moderate-sized coronary thrombus in the chest pain patient. While the initiation of invasive strategies based solely on a RWMA remains controversial, cardiologic consultation, consultative echocardiography, and serial biomarker testing may be prudent. Echocardiographic evaluation of RWMAs, in conjunction with ECG and a targeted history, can be useful in the risk stratification of the ED chest pain patient.
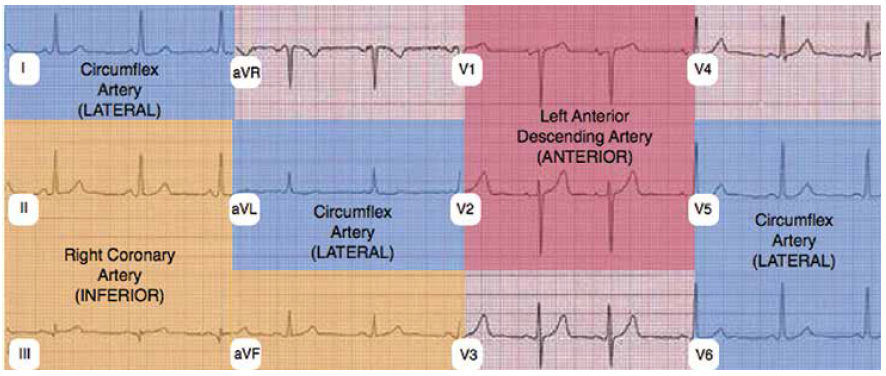
(click for larger image) Figure 4: ECG distribution corresponding to RWMA territories. Anterior RWMA corresponds to left anterior descending artery. Inferior RWMA corresponds to right coronary artery. Lateral RWMA corresponds to circumflex artery.
Considerations, Caveats, and Limitations
When looking for RWMAs with POC echo, there are few notable caveats. Novice sonographers may have difficulty obtaining clear views and should not base decisions on suboptimal imaging. Even for the experienced ED sonographer, differentiating between new and old RMWAs can be extremely challenging. Previous infarctions may result in areas of thinned, akinetic, dyskinetic, or aneurysmal myocardium.15 Also, mechanical and electrical cardiac variants can mimic an acute RWMA. Specifically, focal myocarditis, left bundle branch block, paced rhythms, ventricular aneurysms, Takotsubo cardiomyopathy, and previous cardiac surgeries can all make the acute differentiation of RWMAs difficult. Clinicians performing more advanced echocardiography in the ED should be aware of these and other limitations before adjusting clinical care.
Conclusions
While POC echo to identify RWMAs cannot supplant patient history, clinical examination, ECGs, and cardiac biomarkers, it can provide a prompt bedside tool to help risk-stratify chest pain patients with a risk for myocardial ischemia. Identification of RWMAs may help stratify patients in need of prompt cardiology consultation (and/or comprehensive echocardiography), more frequent serial ECGs, rapid biomarker testing, and in certain cases early cardiac catheterization.
Dr. Johnson and Dr. Lovallo are in the department of emergency medicine at Alameda Health System’s Highland Hospital in Oakland, California.
Dr. Frenkel is in the department of emergency medicine at St. Paul’s Hospital in Vancouver, British Columbia.
Dr. Nagdev is director of emergency ultrasound at Highland Hospital in Oakland, California, and assistant clinical professor (volunteer) of emergency medicine at the University of California, San Francisco.
Anterior Regional Wall Motion Abnormality in a) Parasternal Long Axis and b) Parasternal Short Axis.
Inferior Regional Wall Motion Abnormality in a) Parasternal Short Axis and b) Apical Four Chamber.
Lateral Regional Wall Motion Abnormality in Parasternal Short Axis
References
- McCaig LF, Burt CW. National Hospital Ambulatory Medical Care Survey: 2001 emergency department summary. Adv Data. 2003;(335):1-29.
- Go AS, Mozaffarian D, Roger VL, et al. Heart disease and stroke statistics—2014 update: a report from the American Heart Association. Circulation. 2014;129:e28-92.
- Jneid H, Anderson JL, Wright RS, et al. 2012 ACCF/AHA focused update of the guideline for the management of patients with unstable angina/non–ST-elevation myocardial infarction (updating the 2007 guideline and replacing the 2011 focused update): a report of the American College of Cardiology Foundation/American Heart Association Task Force on practice guidelines. J Am Coll Cardiol. 2012;60:645-681.
- Cheitlin MD, Armstrong WF, Aurigemma GP, et al. ACC/AHA/ASE 2003 guideline update for the clinical application of echocardiography: summary article: a report of the American College of Cardiology/American Heart Association task force on practice guidelines (ACC/AHA/ASE committee to update the 1997 guidelines for the clinical application of echocardiography). Circulation. 2003;108:1146-1162.
- Fleischmann KE, Lee TH, Come PC, et al. Echocardiographic prediction of complications in patients with chest pain. Am J Cardiol. 1997;79:292-298.
- Peels CH, Visser CA, Kupper AJ, et al. Usefulness of two-dimensional echocardiography for immediate detection of myocardial ischemia in the emergency room. Am J Cardiol. 1990;65:687-691.
- Gibson RS, Bishop HL, Stamm RB, et al. Value of early two dimensional echocardiography in patients with acute myocardial infarction. Am J Cardiol. 1982;49:1110-1119.
- Sabia P, Afrookteh A, Touchstone DA, et al. Value of regional wall motion abnormality in the emergency room diagnosis of acute myocardial infarction. A prospective study using two-dimensional echocardiography. Circulation. 1991;84(3 Suppl):I85-92.
- Kontos MC, Arrowood JA, Paulsen WH, et al. Early echocardiography can predict cardiac events in emergency department patients with chest pain. Ann Emerg Med. 1998;31:550-557.
- Kerwin C, Tommaso L, Kulstad E. A brief training module improves recognition of echocardiographic wall-motion abnormalities by emergency medicine physicians. Emerg Med Int. 2011;2011:483242.
- Sobczyk D, Nycz K, Żmudka K. Usefulness of limited echocardiography with A-F mnemonic in patients with suspected non-ST-segment-elevation acute coronary syndrome. Pol Arch Med Wewn. 2014;124:688-694.
- Frenkel O, Riguzzi C, Nagdev A. Identification of high-risk patients with acute coronary syndrome using point-of-care echocardiography in the ED. Am J Emerg Med. 2014;32(6):670–672.
- Ortiz-Pérez JT, Rodríguez J, Meyers SN, et al. Correspondence between the 17-segment model and coronary arterial anatomy using contrast-enhanced cardiac magnetic resonance imaging. JACC Cardiovasc Imaging. 2008;1:282-293.
- Cerqueira MD, Weissman NJ, Dilsizian V, et al. Standardized myocardial segmentation and nomenclature for tomographic imaging of the heart. Circulation. 2002;105:539-542.
- Esmaeilzadeh M, Parsaee M, Maleki M. The role of echocardiography in coronary artery disease and acute myocardial infarction. J Tehran Heart Cent. 2013;8:1-13.
Pages: 1 2 3 4 | Multi-Page




No Responses to “Detect Cardiac Regional Wall Motion Abnormalities by Point-of-Care Echocardiography”