Emergency airways commonly involve challenges of tube placement and oxygenation before and during the procedure. There are a handful of instances, however, when the issue is ventilation and, more specifically, extremes of minute ventilation. Minute ventilation is the amount of air the patient moves in one minute; it is a product of the ventilatory rate and tidal volume (minus dead-space ventilation).
Explore This Issue
ACEP Now: Vol 34 – No 01– January 2015Normal minute ventilation is between 5 and 8 L per minute (Lpm). Tidal volumes of 500 to 600 mL at 12–14 breaths per minute yield minute ventilations between 6.0 and 8.4 L, for example. Minute ventilation can double with light exercise, and it can exceed 40 Lpm with heavy exercise. As defined by the alveolar gas equation, increasing ventilation rate is our body’s only innate mechanism to acutely increase oxygenation. Breathing faster and deeper, we increase the alveolar oxygen tension by decreasing the partial pressure of CO2. Therapeutically, the main method of boosting oxygenation is increasing the inspired oxygen concentration (FiO2). An additional method is to increase the barometric pressure (ie, in a hyperbaric chamber or by rapidly descending from high altitude). Adding positive end-expiratory pressure (PEEP) maintains a pressure in the alveolus throughout the ventilation cycle (and stents open the alveolus, thereby providing more surface area for oxygen absorption).
![Figure 1. A normal minute ventilation involves a minute ventilation between 5 and 8 L [ie, 500–600 mL, rate 10–14 breaths/minute]. In severely ill COPD and asthma patients, overventilation risks auto-PEEP and barotrauma; a starting rate of six breaths with a 500 mL volume allows maximum time for exhalation. Closely monitor blood pressure and vent pressures for auto-PEEP, and adjust up minute ventilation as tolerated. In severely acidotic patients who must maintain a compensatory respiratory alkalosis, match preprocedural minute ventilation during the onset phase of muscle relaxants and after intubation.](https://www.acepnow.com/wp-content/uploads/2015/01/feature-story_pg20-300x179.png)
(click for larger image)
Figure 1.
A normal minute ventilation involves a minute ventilation between 5 and 8 L [ie, 500–600 mL, rate 10–14 breaths/minute]. In severely ill COPD and asthma patients, overventilation risks auto-PEEP and barotrauma; a starting rate of six breaths with a 500 mL volume allows maximum time for exhalation. Closely monitor blood pressure and vent pressures for auto-PEEP, and adjust up minute ventilation as tolerated. In severely acidotic patients who must maintain a compensatory respiratory alkalosis, match preprocedural minute ventilation during the onset phase of muscle relaxants and after intubation.
It’s useful to consider minute ventilation when assessing patients in severe distress. I now appreciate that 15 Lpm via a non-rebreather mask may not meet the minute ventilation of patients in extremis; this explains how a non-rebreather can collapse with inspiration and why many patients feel suffocated with a mask over their face. Patients also do not want to rebreathe their expired CO2, and standard emergency airway equipment, unlike the systems used in the operating room, lacks any CO2 absorption. Nasal oxygen boosts FiO2, flushes the nasopharynx, and fills the upper airway with a high concentration of oxygen available for the next breath. Combining nasal and mask oxygen increases the volume of oxygen available for the patient to inspire. Since I began using nasal oxygen routinely as part of preoxygenation, I have found fewer instances of mask or continuous positive airway pressure (CPAP) intolerance. Even 4–6 Lpm via nasal cannula, which is what I start with in patients who are not critically hypoxic, is very helpful for maximizing pulse oximetry and mask tolerance. I turn nasal cannula up to 15 Lpm after induction and throughout the intubation.
Minute ventilation is not only relevant to pulmonary function and oxygenation, it can affect acute and chronic acid-base balance. Blowing off CO2 increases pH through the conversion of bicarbonate and hydrogen ions to CO2 and H20 (via the carbonic anhydrase reaction). While most ED patients with high minute ventilation have hypoxemia, some patients with respiratory distress have normal pulse oximetry and clear lungs. Their high minute ventilation is to compensate for an underlying severe metabolic acidosis. Compensatory respiratory alkalosis is common in diabetic ketoacidosis, acute renal failure (from rhabdomyolysis and other causes), and salicylate toxicity.
I now appreciate that 15 Lpm via a non-rebreather mask may not meet the minute ventilation of patients in extremis; this explains how a non-rebreather can collapse with inspiration and why many patients feel suffocated with a mask over their face.
At the other extreme of minute ventilation are patients with chronic obstructive pulmonary disease (COPD) and asthma, who have very low minute ventilation. Their problem is not oxygenation and not acid-base related; it’s primarily a problem with air egress. The inability to get air out prevents getting air in, and hyperinflation (breath stacking) results.
I now make minute ventilation a standard part of my airway assessment. In patients who are placed on noninvasive mechanical ventilation, the machine provides the actual minute ventilation directly. I frequently use noninvasive mechanical ventilation as part of preintubation oxygenation in the sickest of patients, especially if PEEP is required. Very high minute ventilation values should trigger suspicion of an acid-base issue. Significant metabolic acidosis (and a compensatory respiratory alkalosis) is usually identified from chemistry values or a blood gas. In my experience, patients with high minute ventilation from respiratory alkalosis often do not have subjective dyspnea, even though their breathing pattern is evidently abnormal to any observer. This is especially true of salicylate poisoning. Severe salicylate poisoning has impressive hyperpnea (excessively deep breathing) with marked tachypnea.
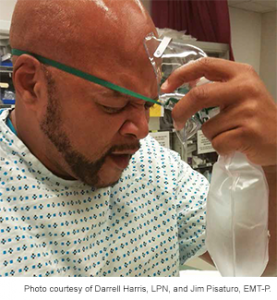
Figure 2.
Think about minute ventilation and oxygenation when patients are intolerant of a non-rebreather. The patients are telling you the flow rate through the mask is not meeting their minute ventilation, and they do not want to re-breathe their CO2. Adding nasal cannula (under the face mask) when treating hypoxemia (and preoxygenating prior to intubation) boosts flow rate, washes out CO2, and significantly increases the effective FiO2.
Normal minute ventilation cannot be used to exclude the presence of severe acidosis, however. Some patients present so fatigued from increased work of breathing when they can no longer compensate; as they tire out and CO2 rises, they become increasingly lethargic. I try to include an elevated CO2 in my differential for lethargy and altered mental status. CO2 retention is easily overlooked, particularly in patients without hypoxia who have baseline mental status problems and limited communication. Hypoxemia is a late manifestation of hypercarbia. Always consider opioid intoxication when patients present with hypoventilation.
We need to be aware of minute ventilation at the extremes because it can cause catastrophic problems in the peri- and postintubation period. Failure to ventilate enough, for instance, failure to maintain respiratory alkalosis in salicylate poisoning (should emergent dialysis not be available prior to intubation), causes worsening of acidosis and has been linked to sudden death. I know of several cases in severe diabetic ketoacidosis where induction and muscle relaxation precipitated death. In hindsight, I think the combination of severe fluid depletion and acute worsening of an already severe acidosis triggered these events. Maintaining a high minute ventilation may have helped the situation.
Conversely, when extreme low minute ventilation is present, operators must be hypervigilant not to create auto-PEEP by breath stacking, which can lead to cardiovascular collapse or barotrauma, resulting in pneumothorax. COPD and asthma patients have as much as a 10 times greater risk of life-threatening hypotension with emergent intubation compared to other ED patients. This results from intubation and over-bagging in a patient with an impaired ability to expel air from the lungs. More volume goes in with each breath than comes out. Hyperinflation of the lungs increases intrathoracic pressure, collapsing the heart, and decreases venous return, leading to hypotension. If not recognized quickly, pulseless electrical activity arrest ensues. The first clue will be a falling pulse oximetry reading, which might be erroneously believed to be a tube problem or barotrauma. In COPD patients (all of whom are at risk of pneumonia), operators also frequently misinterpret postintubation hypotension as a consequence of sepsis instead of auto-PEEP.
Postintubation hypotension should immediately prompt concern for auto-PEEP in COPD and asthma. High plateau pressures will alarm on the vent, and high pressures are felt through the bag, but this may be dismissed as a marker of disease severity. Auto-PEEP is easily corrected by disconnecting the tube from the bag (or vent) and pressing on the chest (pushing air out and suspending ventilation for 30–60 seconds); blood pressure and pulse oximetry will improve quickly.
My goal with every intubation is to avoid a catastrophic event in the peri-intubation and immediate postintubation period. I obsess about oxygenation…I try to avoid regurgitation and emesis.
My goal with every intubation is to avoid a catastrophic event in the peri-intubation and immediate postintubation period. I obsess about oxygenation: preoxygenation with nasal cannulas and face mask or CPAP in the very ill, upright preoxygenation, and NO-DESAT nasal oxygen during all intubations. I try to avoid regurgitation and emesis by avoiding high-pressure face mask ventilation in a flat position, and always positioning the head higher than the stomach (ear-to-sternal notch, or tilting the cervical-spine patient feet down). I decompress the bowel obstructed and massive gastrointestinal bleeders before intubation.
In addition to these techniques that I have adopted for oxygenation and regurgitation prevention, I now try to pick the ventilation strategy that will mimic the patient’s preprocedural minute ventilation. In situations of severe respiratory alkalosis, I aim to re-create the patient’s high minute ventilation after induction and during the onset phase of muscle relaxation (waiting 60 seconds for succinylcholine, or double dose rocuronium or vecuronium, before inserting my direct or video laryngoscope). When bicarbonate has a clear role, (eg, salicylate poisoning, rhabdomyolysis, renal failure with hyperkalemia, etc.), I give it before and sometimes again during induction and intubation. There is controversy about the benefits of sodium bicarbonate in many situations, but when pH is pushed below 7.0, I am liberal with its use, along with maintaining a high minute ventilation.
In the severely ill COPD and asthma patient, I start with a simple ventilation strategy for postintubation: six breaths a minute and 500 cc tidal volume (3 L minute ventilation). By deliberately going slow, I allow the maximal time between breaths for air to get out. I check plateau pressures on the vent and blood pressure. I aim to increase minute ventilation, but I do so slowly, making sure not to trigger auto-PEEP and hypotension. In addition to nebulized bronchodilators, you can also add intravenous ketamine, postintubation, for both bronchodilation and sedation. The 3 L minute ventilation of 500 cc/6 breaths will not correct the CO2 retention, but it is safest to correct this slowly as the patient’s pulmonary function allows (watching vent pressures and blood pressure). “Permissive hypercapnia” is the deliberate strategy of not correcting the CO2 quickly; a bicarbonate drip may be added if acidosis drops the pH below 7, but have not been proved to improve outcomes.
For the vast majority of ED airways, ventilation is not a major concern. Use a strategy of relatively low volume (6–7 mL/kg) and relatively low rates with low pressures (and gentle inflation); the goal is to avoid overinflation, gastric distention, and regurgitation. When the patient presents at the extremes of ventilation, however, remember that plastic in trachea is not the primary solution. If intubation cannot be avoided, aim to re-create the immediate preintubation minute ventilation while bagging during the onset of muscle relaxation and immediately postintubation. By recognizing patients with a compensatory respiratory alkalosis, and the very low minute ventilation of COPD and asthma patients, you will avoid precipitating a peri-intubation catastrophe.
 Dr. Levitan is an adjunct professor of emergency medicine at Dartmouth College’s Geisel School of Medicine in Hanover, N.H., and a visiting professor of emergency medicine at the University of Maryland in Baltimore. He works clinically at critical care access hospitals in rural New Hampshire and teaches cadaveric and fiber-optic airway courses.
Dr. Levitan is an adjunct professor of emergency medicine at Dartmouth College’s Geisel School of Medicine in Hanover, N.H., and a visiting professor of emergency medicine at the University of Maryland in Baltimore. He works clinically at critical care access hospitals in rural New Hampshire and teaches cadaveric and fiber-optic airway courses.





One Response to “Avoid Airway Catastrophes on the Extremes of Minute Ventilation”
January 16, 2021
Dr. onkar ThakurVery informative and practical, Thanks to Dr. Richard. Please keep me posted.